My experiment
1. purpose
Polymeric materials have remarkably developed and become indispensable to everyday life. In particular, recently, biodegradable polymer which is friendly for environment has attracted attention. In this work, we studied the poly (γ-glutamic acid)(γ-PGA) by solid state NMR its structure.
2. Introduction
NMR spectroscopy is an effective analysis method in the structural analysis. It is obtained information on individual atomic level that makes up the molecule. This spectroscopy exerts power in structure determination. The following three examples are pointed out as the nature for analytical chemistry of this spectroscopy. It is comprehension, selectivity and non destruction. That is, by this spectroscopy, without destroying the sample, whole image or detail of the sample or unknown sample can be grasped. The following three examples are pointed out as the nature for organic chemistry of this spectroscopy. It is structure determination of unknown compound, partial structure determination and identification of known compound. On the other hand, it has demerits. For example, low sensitivity and problem of measurement and time. NMR spectroscopy is low sensitivity compared to analytical methods capable of sensitive detection, such as chromatography or mass spectroscopy, and measurement of the detection of the reaction intermediates and unstable compounds is difficult.
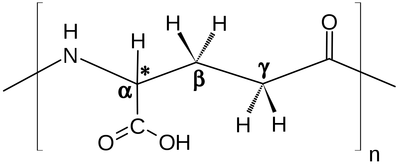
Polyglutamic acid (PGA) is a polymer of the amino acid glutamic acid (GA). Poly-γ-glutamic acid(γ-PGA) (the form where the peptide bonds are between the amino group of GA and the carboxyl group at the end of the GA side chain) is a major constituent of the Japanese food natto. γ-PGA has a wide number of potential uses ranging from food and medicine to water treatment. It is also widely being used as a drug delivery system in cancer treatment.
3. Result and discussion
Carbonyl carbon peaks in 13C solid NMR spectrum of γ-PGA powder differs from that ofγ-PGA /Na powder. γ-PGA shows an additional sharp peak at 171 ppm. Carbonyl carbon peak ofγ-PGA was deconvoluted into three peaks: Two peaks in the higher field side were assigned to two different kinds of side-chain carboxyl groups, one for the carboxyl group which forms the strong hydrogen bonds among carboxyl groups (dimeric form) and the other for free carboxyl group. A lowest field side peak was assigned to carbonyl carbon of main chain amide linkage. On the other hand carbonyl carbon peak ofγ-PGA/Na was deconvoluted into two peaks: one for main chain amide linkage and the other for free carboxyl group. 13C solid NMR spectra of DL-γ-PGA with different D / L ratio show almost the same form in carbonyl carbon region. But 13C solid NMR spectra of only L-γ-PGA doesn’t show a peak of 171 ppm considered to be a dimer. My future work is to consider why this peak does not appear in L-γ-PGA.
Polymeric materials have remarkably developed and become indispensable to everyday life. In particular, recently, biodegradable polymer which is friendly for environment has attracted attention. In this work, we studied the poly (γ-glutamic acid)(γ-PGA) by solid state NMR its structure.
2. Introduction
NMR spectroscopy is an effective analysis method in the structural analysis. It is obtained information on individual atomic level that makes up the molecule. This spectroscopy exerts power in structure determination. The following three examples are pointed out as the nature for analytical chemistry of this spectroscopy. It is comprehension, selectivity and non destruction. That is, by this spectroscopy, without destroying the sample, whole image or detail of the sample or unknown sample can be grasped. The following three examples are pointed out as the nature for organic chemistry of this spectroscopy. It is structure determination of unknown compound, partial structure determination and identification of known compound. On the other hand, it has demerits. For example, low sensitivity and problem of measurement and time. NMR spectroscopy is low sensitivity compared to analytical methods capable of sensitive detection, such as chromatography or mass spectroscopy, and measurement of the detection of the reaction intermediates and unstable compounds is difficult.
Polyglutamic acid (PGA) is a polymer of the amino acid glutamic acid (GA). Poly-γ-glutamic acid(γ-PGA) (the form where the peptide bonds are between the amino group of GA and the carboxyl group at the end of the GA side chain) is a major constituent of the Japanese food natto. γ-PGA has a wide number of potential uses ranging from food and medicine to water treatment. It is also widely being used as a drug delivery system in cancer treatment.
3. Result and discussion
Carbonyl carbon peaks in 13C solid NMR spectrum of γ-PGA powder differs from that ofγ-PGA /Na powder. γ-PGA shows an additional sharp peak at 171 ppm. Carbonyl carbon peak ofγ-PGA was deconvoluted into three peaks: Two peaks in the higher field side were assigned to two different kinds of side-chain carboxyl groups, one for the carboxyl group which forms the strong hydrogen bonds among carboxyl groups (dimeric form) and the other for free carboxyl group. A lowest field side peak was assigned to carbonyl carbon of main chain amide linkage. On the other hand carbonyl carbon peak ofγ-PGA/Na was deconvoluted into two peaks: one for main chain amide linkage and the other for free carboxyl group. 13C solid NMR spectra of DL-γ-PGA with different D / L ratio show almost the same form in carbonyl carbon region. But 13C solid NMR spectra of only L-γ-PGA doesn’t show a peak of 171 ppm considered to be a dimer. My future work is to consider why this peak does not appear in L-γ-PGA.
